Photo Credit: Judd Case
Sandwich Bluff on Vega Island, northern Antarctic Peninsula, where the Vegavis specimen and many other late Cretaceous, Antarctic bird specimens, have been recovered. The specimens here range in age from 70 to 67.5 million years ago.
Antarctic bird nest?
Discovery of avian fossils suggests Antarctica may have been origin of modern species
By Peter Rejcek, Antarctic Sun Editor
Posted June 5, 2009
Julia Clarke External Non-U.S. government site has good reason to believe fossils collected from islands along the Antarctic Peninsula could yield new insights into the evolutionary history of modern birds.
After all, about five years ago, she and her U.S. and Argentine colleagues found proof from a rock specimen, which contained avian vertebrae and pelvic bones among other bits of skeleton, that close relatives of at least one order of modern birds co-existed with dinosaurs.
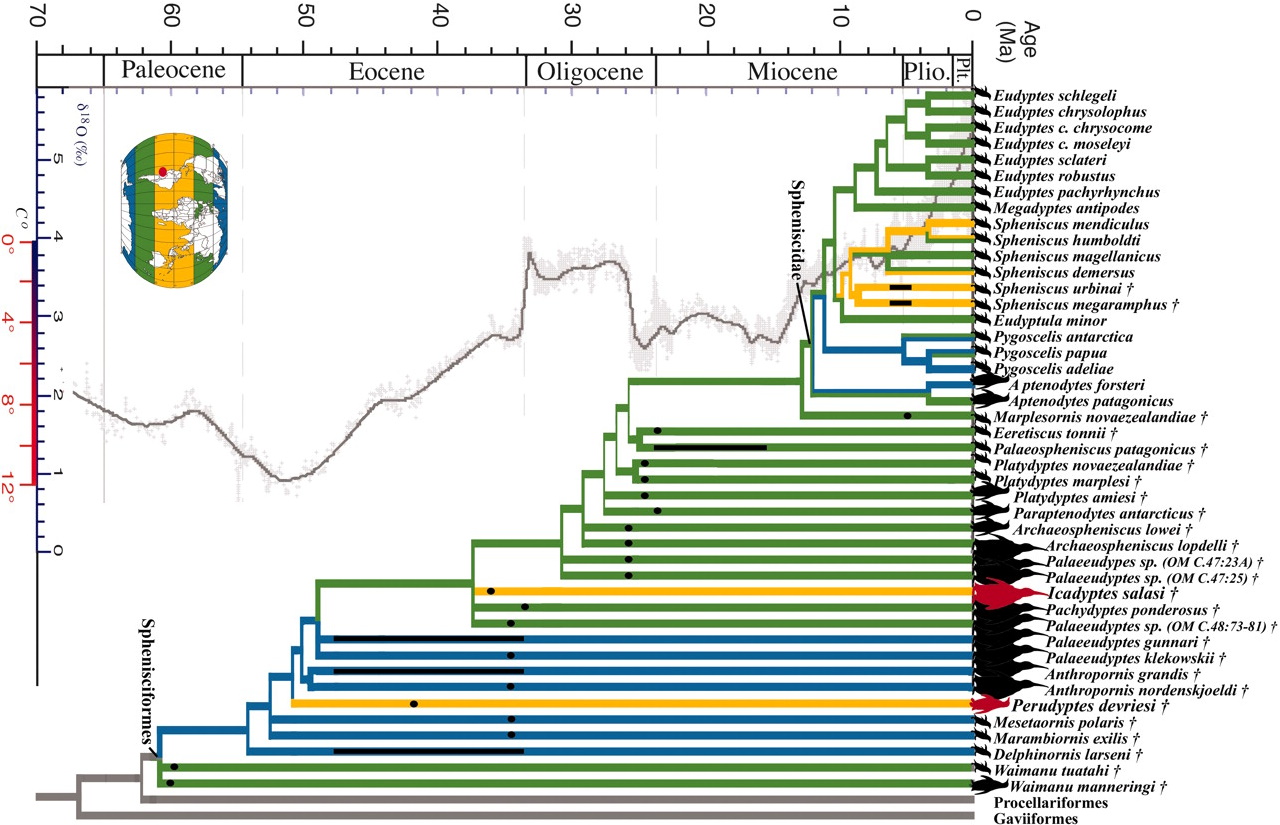
The discovery of the new species, Vegavis iaai, collected in 1992 by scientists from Argentina on Vega Island and re-examined by Clarke and her U.S. and Argentine team more than a decade later, using high-resolution X-ray computed tomography (CT) technology, resulted in a paper in the journal Nature in 2005. Based on the data, Vegavis iaai fell within the order Anseriformes, which includes ducks, geese and swan.
Vegavis lived more than 65 million years ago, well before Antarctica turned into an icehouse. It also existed just before the mass extinction that swept the dinosaurs from the Earth along with up to three-quarters of all species.
The find was important because it offered some of the best fossil evidence to date that linked modern bird divergence, the spread of today’s species, before the K-T boundary — when geologic time turned the page from the Cretaceous to the Tertiary period after the mass extinction.
On one side of the controversy are scientists who argue that molecular evidence and modern distribution of living bird groups suggest that their ancestors existed alongside non-avian dinosaurs well before the K-T extinction, perhaps tens of millions of years earlier. Others have claimed the fossil record shows no real evidence of living bird lineages in the Cretaceous. Only after the extinction, they say, did modern bird evolution take flight.
“It’s still really extraordinarily contentious five years later what lineages are present in the Cretaceous prior to the K-T boundary,” said Clarke, associate professor in the Department of Geological Sciences at the University of Texas at Austin External Non-U.S. government site. “We propose you have the beginnings of this radiation supported [by the Vegavis specimen]. We don’t see any evidence yet of [all extant bird radiation] having happened so much earlier in the fossil record.”
Clarke and her colleagues believe they may find additional information on what lineages date before the K-T boundary by examining additional fossils collected from Antarctica over the last 20 years.
Growing evidence
Clarke is the principal investigator (PI) on a Small Grant for Exploratory Research (SGER) from the National Science Foundation (NSF) External U.S. government site to pull together the data and experts from the United States and Argentina to see what the fossil record from the Antarctic Peninsula says about the evolutionary history of other orders of birds, most of which fall into the superorder Neoaves. (It was a separate SGER project that led to the discovery that Vegavis was closely related to modern waterfowl, Anseriformes, which fall into the superorder Galloanserae.)

Julia Clarke
Photo Credit: North Carolina State University
Julia Clarke holds a penguin skull related to separate avian fossil research.
“The partial skeletons … from Antarctica are the best candidates for being part of that extant radiation,” Clarke said. “The nice thing about getting this team together is we’re sharing information, we’re sharing data, and hopefully coming to a consensus view of what diversity is represented,” Clarke said.
Her co-PI on the project is Judd Case, dean of Eastern Washington University’s College of Science, Health and Engineering External Non-U.S. government site. He made several expeditions to Vega and James Ross islands between 1997 and 2004 on the northern tip of the Antarctic Peninsula to hunt for vertebrate fossils. The goal of those expeditions was to put together a more complete picture of dinosaur, mammal and reptile geographic distribution and evolution between 80 and 65 million years ago.
Case said the material from those finds has further bolstered the hypothesis of the earlier radiation of modern birds, with at least four lineages emerging before the K-T extinction based on the evidence the scientists are assembling.
“We’ve got a good amount of material to make the judgment,” he said. “We’ve got good documented, late Cretaceous deposits,” including specimens from the major clade, or group, Neoaves.
“The fossil data, along with the molecular data, continue to point to this older origination, and it’s providing a location where this … may have occurred,” Case said.
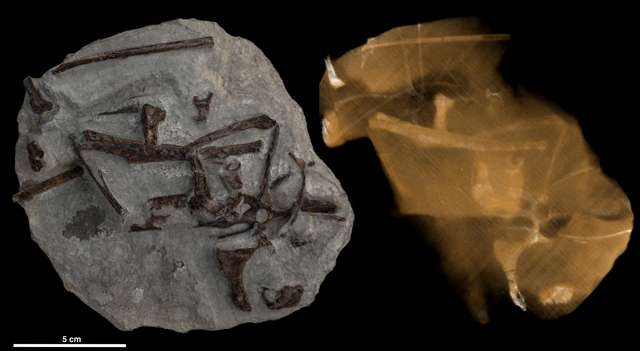 Ground zero for bird origination
Ground zero for bird originationVegavis iaai concretion, left, and the CT scan of the rock and bird fossils. CT scans offer a noninvasive way for peering into solid objects and for obtaining digital information on their 3-D geometries and properties.
In fact, the paleontologists believe this region may have been ground zero for modern bird evolution, or at least a major hotspot. The hypothesis is based largely on the biogeography, the distribution of existing bird groups, an idea that dates back to the 1970s, according to Clarke.
“That idea has been around for a long time,” she said, but the fossil evidence hasn’t been collected and evaluated in a systematic fashion, as she and her team proposes to do, to put it to the test.
Case said evidence is mounting for such a scenario, especially given the Southern Hemisphere origination of lineages for penguins and ratites (such as ostriches and emus). “If you throw those lines in with [the other pre-K-T boundary lineages], it clearly begins to allow for the hypothesis, and add some strength to the idea, that somewhere deep in Gondwana, modern birds originated.”
Gondwana is the southern supercontinent that at one time included Antarctica, South America, Africa, Madagascar, Australia-New Guinea and New Zealand. By the late Cretaceous, Antarctica had moved into its current position, though still tenuously connected to Australia and South America.
The climate would have been quite moderate, according to Case. “Where today [Antarctica] seems pretty stark … back in this timeframe, it was a very rich, vibrant place,” he said. “The contrast of what it was to what it is today is pretty dramatic. I can’t think any place else on the Earth as dichotomous.
“We’re looking at cool, temperate waters, certainly not much different than what we see off the west coast of the United States, at least until you get to southern California,” he added. “We don’t expect marine reptiles to have a lot of blubber, like marine mammals do, so it’s got to be warm enough that the young with small body sizes can survive comfortably.”
Clarke said that while there appears to be good evidence emerging from the Antarctic Peninsula fossil record to suggest a Southern Hemisphere origination, she cautioned that the sampling size is still relatively small.
“We’re going to be able to be able to offer more data than has ever been put forward on what’s really in Antarctica during this time period, but we’re not going to be able to answer this question definitively,” she said.
Breaking rocks digitally
Clarke’s expertise is phylogenetic analysis from morphological data, meaning she studies the evolutionary relatedness among birds by comparing the physical changes that occur over time. A very useful tool in acquiring the data she needs for the research is a high-resolution X-ray CT scanner.
CT scans offer a noninvasive way for peering into solid objects and for obtaining digital information on their 3-D geometries and properties. High-resolution X-ray CT differs from conventional medical CAT-scanning in its ability to provide details down to the tens of microns — less than the width of a hair. The NSF supports the High-Resolution X-ray Computed Tomography Facility External Non-U.S. government site at the University of Texas at Austin (UTCT).
Clarke said the scans save time and allow scientists to manipulate the data in new ways. “CT scans are really useful in this case because a lot of the rocks in which these remains are preserved are incredibly hard,” she explained. “It’s months and months and months of moving individual sand grains to prepare these things. You can see the interiors of elements. You could see morphologies that would never be exposed even if you didn't have those tools.
“There were bones that were discovered in that Vegavis specimen that we had no idea were in there,” she added. In addition, one of her students is digitally extracting individual bones from the original Vegavis scan, which appears online like the silvery half of an oversized grapefruit, with the ancient bird bones prominently sticking out.
Work has already begun on the fossils from Argentina, Texas Tech, the South Dakota School of Mines and elsewhere. Eventually, the team plans to publish a monograph, a sort of comprehensive essay, which describes the Antarctic materials, with a photo library of all the remains with preliminary identifications, according to Clarke.
“We’re making progress,” she said.
Case said the researchers would propose further fieldwork to look specifically at bird fossils and the question of whether that part of Gondwana was indeed the cradle for modern birds.
“It’s one of the biggest questions out there right now,” he said.
NSF-funded research in this story: Julia Clarke, University of Texas at Austin, and Judd Case, Eastern Washington University, Award No. 0731404 External U.S. government site.
Source:
http://antarcticsun.usap.gov/science/contentHandler.cfm?id=1785